Our website uses cookies so that we can provide you with the best user experience possible. Cookie information is stored in your browser and performs functions such as recognising you when you return to our website and helping our team to understand which sections of the website you find most interesting and useful. Thank you!
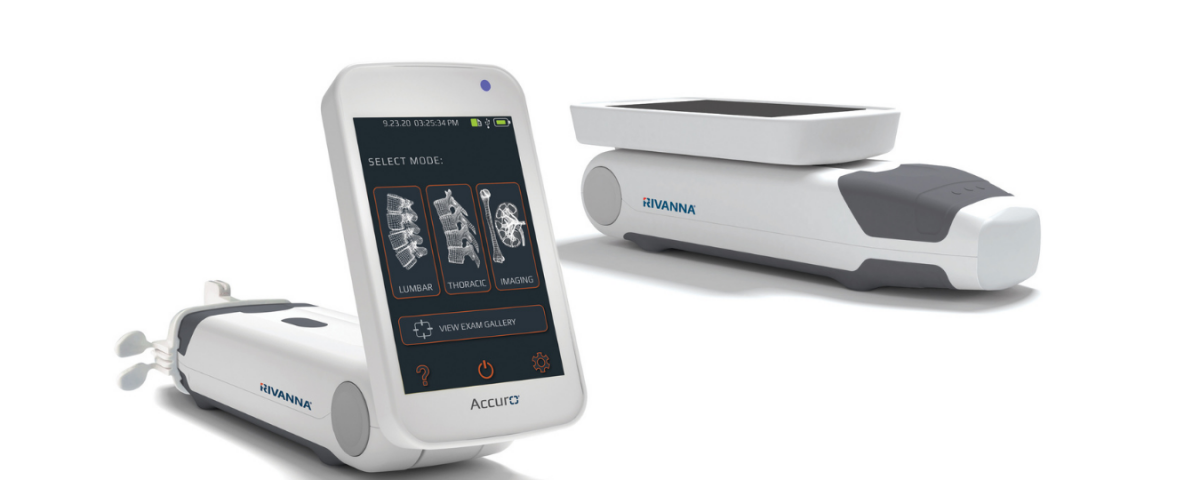
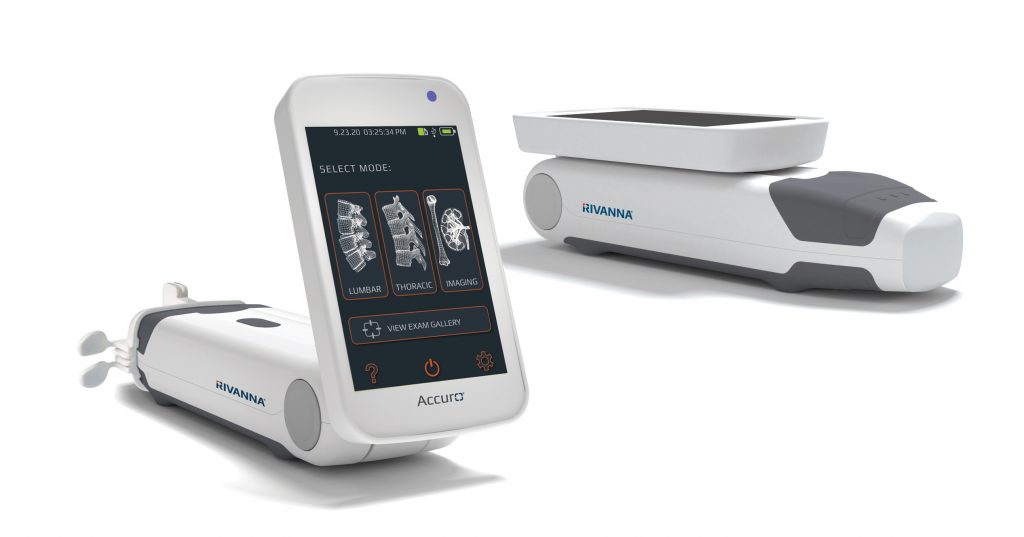
CHARLOTTESVILLE, VA, July 13, 2021 — RIVANNA®, developers of world-first, imaging-based medical solutions, today announced sales of the Accuro device and related service and equipment increased 263 percent versus the same period of the prior year following second-quarter results ending June 30, 2021. International sales generated 73 percent of the sales revenue, indicating the success of RIVANNA’s expanding global distribution footprint.
“We are pleased with our second-quarter sales results, which reflect Accuro’s value to its customers and signifies a migration away from palpation-based needle placement in favor of a safer and more efficient image-guided standard for spinal needle placement,” commented Danielle Faulk, National Accounts Manager. “Currently, there are no modalities like Accuro for anesthesia providers, specifically designed to improve the efficiency of spinal needle guidance procedures.”
Accuro lumbar and thoracic presets, with SpineNav3D™ AI-Enabled Image Recognition technology, facilitate image interpretation of individual 2D lumbar and thoracic spine scans by automating spinal bone landmark detection and depth measurements and providing a real-time assessment of scan plane orientation in 3D. This technology makes image interpretation and measurements of the lumbar and thoracic spine anatomy simple and accessible.
Will Mauldin, PhD, Co-Founder, and CEO of RIVANNA, commented, “Second-quarter Accuro sales exceeded our expectations and were due in part to our strategic distribution partnerships. I’m thrilled to report that following an unpredictable year through the pandemic, the RIVANNA team has forged ahead with great resolve.” Mr. Mauldin continued, “With the increasing need for our products and technologies, we have expanded our manufacturing capacity to meet the growing global demand for the Accuro product line and related medical equipment and components. We are well-positioned to accelerate progress on our next-generation product platforms and continue to advance our mission to elevate the global standard of care.”
The product-line expansion, including Accuro 3S and Accuro XV, builds on RIVANNA’s groundbreaking ultrasound-based imaging technologies. BoneVision™ and BoneEnhance® optimize ultrasound image acquisition and visualization to provide radiation-free alternatives to X-ray-based imaging modalities. When paired with SpineNav3D™ AI-Enabled Image Recognition, these products offer comprehensive clinician-assistance solutions that elevate the standard of care across a range of clinical applications.
Commercialization of the Accuro 3S and Accuro XV will address a billion-dollar market opportunity in pain management, neurology, oncology and emergency medicine.
ABOUT RIVANNA
RIVANNA is elevating global standards of care through the development and commercialization of world-first imaging-based medical technologies, including BoneVision™ Multi-Probe Multi-Angle Image Acquisition, BoneEnhance® Multi-Frequency Image Reconstruction and SpineNav3D™ AI-Enabled Image Recognition. RIVANNA’s complete clinician-assistance solutions improve decision-making, workflows, health outcomes and patient satisfaction. Headquartered in Charlottesville, VA, RIVANNA is privately held and operates an FDA-registered and ISO 13485:2016 certified manufacturing facility. For more information, visit rivannamedical.com.