For RIVANNA, AI can help clinicians metaphorically see through walls by layering information on top of ultrasound to achieve X-ray-like performance without radiation. Co-founded in 2010 by CEO Will Mauldin, PhD, the company emerged when the first handheld ultrasound devices were emerging onto the market during a large-scale push for miniaturized electronics. Mauldin, who specialized in ultrasound technologies as a doctoral candidate and later served as an assistant research professor at UVA, saw that trend as an opportunity to address an unmet need in spinal anesthesia, particularly for childbirth. Since X-rays cannot be used around a fetus, epidural needles are placed without any image guidance, entailing serious risks.
The bony environment of the spine and ribcage is obstructive to ultrasound imaging, a modality more suited for soft tissue and one that anesthesiologists only use in limited contexts. This anatomical challenge has traditionally made real-time visualization of the anesthesia needle entering the epidural space impossible when clinicians are used to seeing the entire path of the needle to its target when administering vascular access and regional anesthesia. However, more recent advancements in ultrasound probe technology have given way to a safer, albeit more painstaking, guided methodology. Managing the positioning of the probe and needle so that one doesn’t block the other is a three-handed task, as the anesthesiologist needs to handle both the needle and syringe separately, thus requiring a second personnel member for the job.
Spinal, or neuraxial, anesthesia is only effective if it is delivered to the right location, the epidural space. According to Mauldin, up to 1 in 5 patients in labor go without pain relief due to inaccurate placement. Plus, multiple attempts to access the epidural space can traumatize the peripheral nerves near the spine, causing paresthesia. In non-obstetric applications such as lower limb surgeries, doctors may need to opt for general anesthesia over neuraxial, which carries its own risk factors. Patients with spinal implants or scar tissue from previous injuries or surgery may be contraindicated for epidurals altogether if needle access is too complicated.
Because the epidural space is not a solid structure, but a region beyond the ligamentum flavum in the posterior complex adjacent to the dura, anesthesiologists use a “loss of resistance” technique to determine when the needle pierces the ligament into the space and stop advancing before reaching the dura. If the needle extends too far and punctures the dura of the spinal cord, debilitating headaches can result from a leak of spinal fluid that requires a separate procedure to resolve and might lead to lasting consequences downstream. With nearly 5 million epidural injections performed annually across obstetric, surgical, and chronic pain settings, “even though these are low single-digit rates, we’re still talking about tens of thousands of patients left with chronic neurological conditions,” Mauldin says.
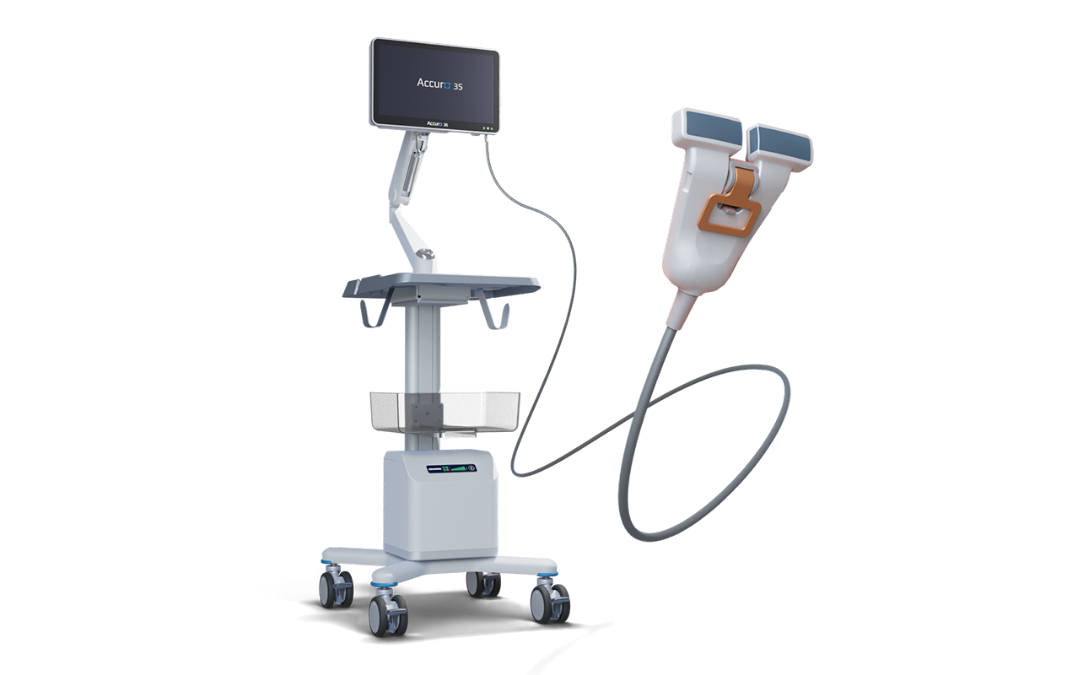
The CEO describes RIVANNA’s debut device Accuro as “a stud finder for the spine.” Using relatively basic AI developed in the 2010s for enhanced signal processing and bone mapping, Accuro provided physicians with a view of the needle’s position relative to the epidural space using B-MODE ultrasound. The second generation, Accuro 3S, started development in 2021, was FDA-cleared in 2025, and is slated for a limited market release toward the end of this year. The updated model has more powerful AI that provides “a true, automated, real-time image guided technique” according to Mauldin and a modified design to solve the three-hand problem without the need for a second clinician.
As a member of the 2025 MedTech Innovator cohort and a top-five finalist for the Grand Prize at The MedTech Conference powered by AdvaMed, RIVANNA has received vital exposure to industry leaders and stakeholders. CEO Will Mauldin notes, “One big benefit we’ve gotten from the relationship with MTI has been the connections to resources that were instrumental in forming strategic partnerships, raising money, and commercialization,” adding that participation in MTI’s investor forum at the JP Morgan conference earlier this year has brought the company closer into the fold of the overall medtech ecosystem.
When anesthesiologists place a central line using ultrasound guidance, their targets are fairly shallow, within a few centimeters of the skin surface. The needle is placed at an oblique angle to the probe that can’t be replicated with epidural procedures since the needle and probe must both be perpendicular to the access point. To make this configuration possible, Accuro 3S has an opening in the center of a patented Dual-Array probe that the needle can pass through without disrupting the field of view or compromising the image. This innovation addresses key challenges in current procedural guidance, and the company recognizes the foundational research of Dr. Charles Y. Kim (Duke University), whose work in ultrasound-guided interventions was instrumental in this advancement.
The core IP behind Accuro and its underlying software SpineNav3D is exclusively licensed from the University of Virginia, and while the first iteration included a disposable attachment that made a temporary marking on the patient’s skin indicating the alignment of the needle, the Accuro 3S system replaces that component with a different consumable that secures the probe to the patient’s back.
Compared with the “rules-based” AI found in Accuro, Accuro 3S incorporates SpineNav-AI, a more robust neural network that Mauldin says is “much more performant in terms of raw accuracy” and conveys more annotated information, including locations of the ligament, spinal canal, muscle tissue, and more. Since spinal ultrasound data was not abundant, RIVANNA had to collect its own algorithm training data prospectively, gathering between 10,000 and 15,000 scans of patients ranging in age, sex, ethnicity, and body mass index.
SpineNav-AI software is included in the 510(k) clearance of Accuro 3S as a diagnostic ultrasound system as opposed to being regulated as a standalone medical device because it is not intended for pairing with any other ultrasound hardware. Still, the algorithm requires an additional battery of clinical testing for validation, and Mauldin is not opposed to the possibility of licensing it as an independent product.
“There’s a compelling business case to leverage [the software] that we’ve created, and to deploy it on a leading point-of-care ultrasound manufacturer’s system,” Mauldin states. “One segment of the market prefers an all-in-one solution, but we also know that emergency medicine constitutes a large segment of the market that does a lot of lumbar punctures and POC ultrasound scans that would value the technology on the devices they’re already familiar with.” The CEO also anticipates opportunities for future applications in thoracic epidurals, paravertebral blocks, and chronic pain solutions that tend to use more capital-intensive and radiation-requiring fluoroscopy.
Reviewed by an independent panel of radiologists, SpineNav3D— the foundational algorithm powering the first-generation Accuro and predecessor to SpineNav-AI—proved to be more than 94% accurate relative to the ground truth within 3mm. Clinical trials comparing the Accuro system with epidurals performed without guidance demonstrated an 81% higher first-insertion success rate, 83% fewer instances of paresthesia, and a 90% reduction in patients reporting a satisfaction score of less than 4 out of 5 with their pain management experience.
In addition to being able to utilize an existing CPT code that Mauldin says is “uniquely applicable” to Accuro 3S procedures, RIVANNA is also in communication with CMS regarding a new ICD-10-PCS code that will help with tracking healthcare economics and outcomes when using the devices. The company has raised a total of $9.5 million through its Series B, primarily from family office shareholders, and has received $52 million in cumulative funding from BARDA under a contract to advance its musculoskeletal imaging platform for fracture detection and soft tissue diagnostics, unrelated to its epidural product line.
With FDA clearance secured, an existing reimbursement pathway, and a licensable AI platform that could reach well beyond its own hardware, Mauldin sees RIVANNA at a commercial inflection point: “We’ve de-risked the technology—now it’s about scaling it across a market where millions of procedures are still performed blind every year.”