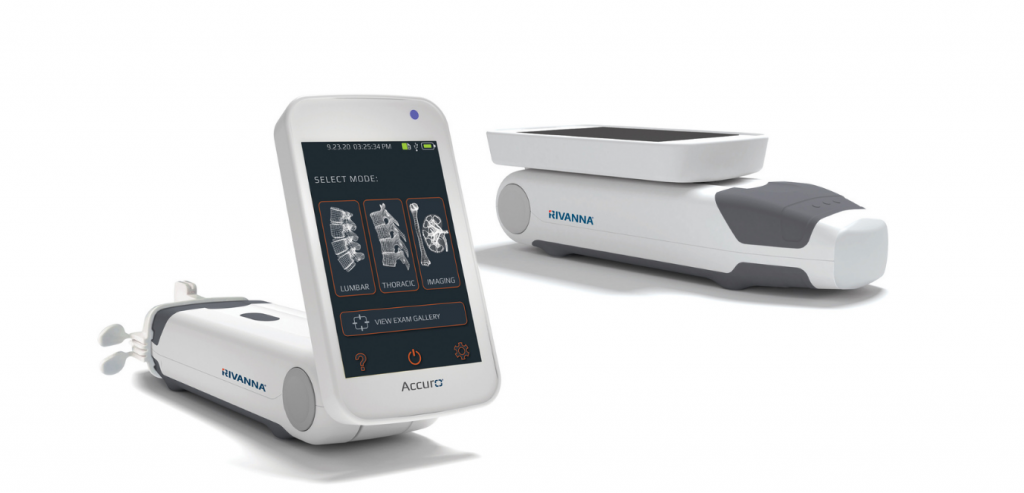
RIVANNA Receives FDA 510(k) Clearance for Accuro XV Musculoskeletal Ultrasound Imaging System
CHARLOTTESVILLE, VA – MAY 7, 2026 – RIVANNA®, developer of AI-enabled clinical decision-support solutions, today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for the Accuro® XV Diagnostic Ultrasound System. The clearance...